Design Exquisite Molecules that are Makeable
Chemify’s Quantum Chemputation enables unbiased, synthesis-aware exploration of chemical space, directly producing real molecules with superior design, optimized properties, and synthetic tractability.
Context
Chemify and Lgenia, supported by the Gates Foundation, are developing novel small-molecule inhibitors of Mycobacterium tuberculosis NAD synthetase (Mtb NadE) with improved drug-like properties. NadE, the final enzyme in NAD biosynthesis, is essential for Mtb energy and redox balance; its inhibition depletes NAD and rapidly kills both active and dormant bacteria, offering a powerful route to shorten TB treatment and overcome persistence.
Approach
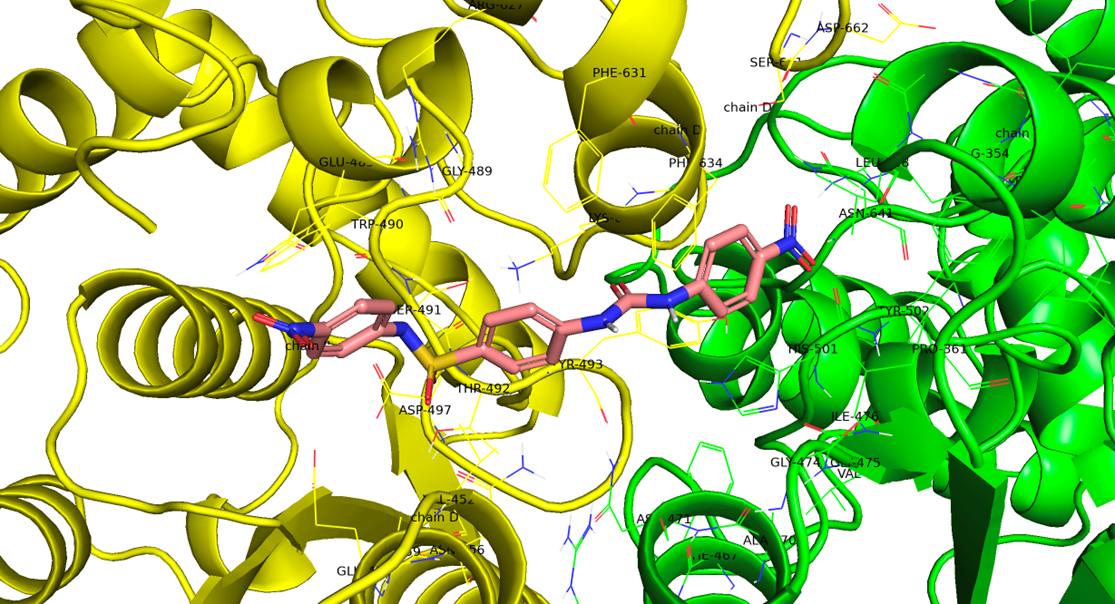
Lgenia provided a set of known Mtb NadE inhibitors (Wang series), which, despite confirmed activity, suffer from poor solubility and permeability. In Cycle 1, Chemify synthesized the 12 original Wang compounds and designed 28 analogues to map key pharmacophoric features. Using docking and molecular dynamics with the high-resolution PDB 6OFC structure, Chemify determined the likely bioactive conformation of the lead Compound 4f and developed a refined binding model. Protein–ligand interactions were validated via PLIP, with MD simulations revealing critical contacts within the NadE pocket.
Initial results identified key pharmacophores, validated through ligand-based modelling. The model was benchmarked with SAR data using ROC/PR analysis to define selection thresholds and guide design of new active molecules.
Cycle 1 (Wang and extended Wang compounds)
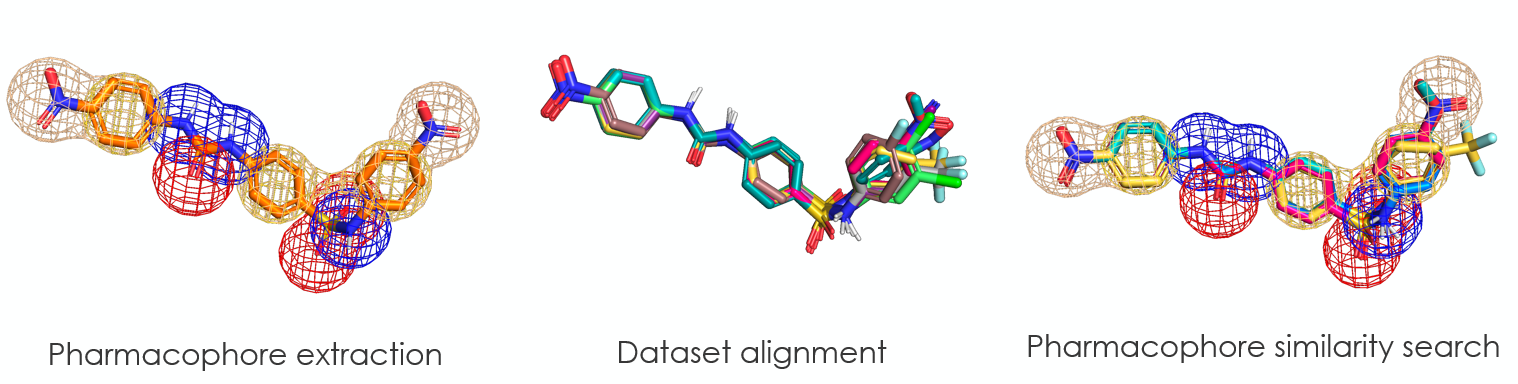
Cycles 2 and 3 (Chemify Designs)
Building on Cycle 1, Chemify used Quantum Chemputation to generate over 786 million virtual, synthesizable compounds. Guided by Cycle 1 pharmacophore insights (4f similarity, LogP 1–4, MW 200–500), 2D filtering reduced this to ~50,000 candidates, followed by 3D ligand- and structure-based ranking. Fourteen core scaffolds and 70 compounds were synthesized across Cycles 2–3, accessing highly novel chemical space with improved drug-like properties.


Results
To date, we have designed, synthesized, and tested novel Mtb NadE inhibitors with markedly improved permeability and solubility over known compounds.
Differentiating Features
Our Quantum Chemputation technology explores novel chemical space in which every design is synthetically tractable. It expands access to unexplored regions of chemistry while bridging the gap between design and synthesis. This enables deeper exploration and direct realization of new molecules with superior design quality, optimized properties, and proven synthetic feasibility.
More Case Studies
Chemputability – Upfront Makeability Assessment
Chemify’s technology algorithmically scores each molecule’s Chemputability—its practical makeability—before synthesis begins. This upfront assessment reveals whether a design can be made and, if not, it suggests Chemputable alternatives, bridging the gap between design and make.
Massive Library Derivatization Deep into Chemical Space
Once a synthetic route is established—even 15+ steps—our technology can repeatedly access and explore that region of chemical space, generating vast derivatized libraries for deep discovery.
Let's Work Together
Collaboration with Chemify Starts Here
If you would like to discover novel molecules with superior outcomes that are makeable, we would love to hear from you.


